The Center provides customized usability consultations across the product lifecycle — from initial concept to design to implementation — to meet users’ critical needs as digital health and innovative technology rapidly advance.
Our experts test the safety and effectiveness of products, providing valuable insight to ensure they are market-ready. Because of our unique setting within MedStar Health, the Center has unparalleled access to conduct usability tests in an actual clinical environment (in-situ testing).
In addition, MedStar Health’s extensive simulation centers provide a variety of test settings, including high-fidelity, patient-based healthcare simulators, “serious game-based” simulations, and others.
We have experience with:
-
Medical devices and combination products in all healthcare domains
-
Medical device accessories
-
Websites and mobile applications
-
Health IT (electronic health records, telehealth, etc.)
-
Home-use products
-
Training programs
-
Instructions for use
-
Signage and trainings
Examples of our approach at every stage of product development:
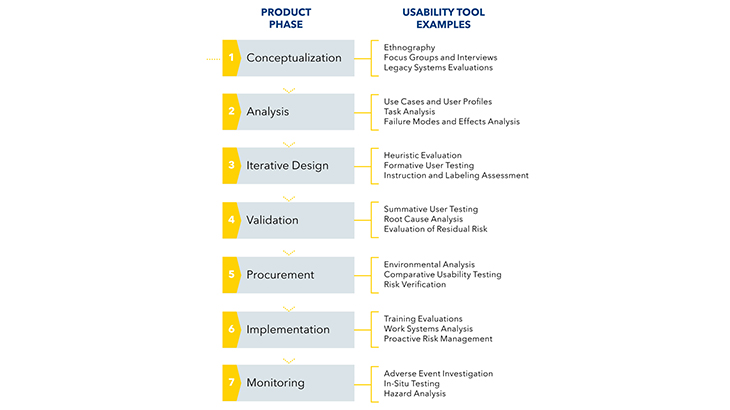
Usability evaluation
Digital health is revolutionizing healthcare and making the intersection of technology and human interaction more essential than ever. It will take ongoing support and investment — backed by the right skills and knowledge — to better design and implement new processes and systems. With growth in digital health, the role of human factors is imperative to the safety of the technology and the patient and clinician experience.
Guidance for FDA regulatory submission
Our usability experts are continually working with the FDA and staying current on evolving requirements. We conduct evaluations for application of human factors to the design and development of medical devices and products to ensure compliance with FDA requirements. From assessing user needs, risk evaluation, and critical task identification, to formative and validation stage user testing, our processes are designed to comply with the standards for FDA submission.
As examples, our evaluations have pertained to the following regulations:
-
FDA Human Factors Guidance for 510(k) Pre-Market Approval
-
ANSI/AAMI HE75 Human Factors Engineering – Design of Medical Devices
-
ISO 62366 – Application of Usability Engineering to Medical Devices
-
ISO 9241 – Ergonomics of Human System Interaction
-
Other relevant medical application standards such as MIL, W3C, etc.
Additional information
Center overview
Read the MedStar Human Factors Center Overview for the most recent summary of our work.









